Florham Park, NJ, November 9th, 2020
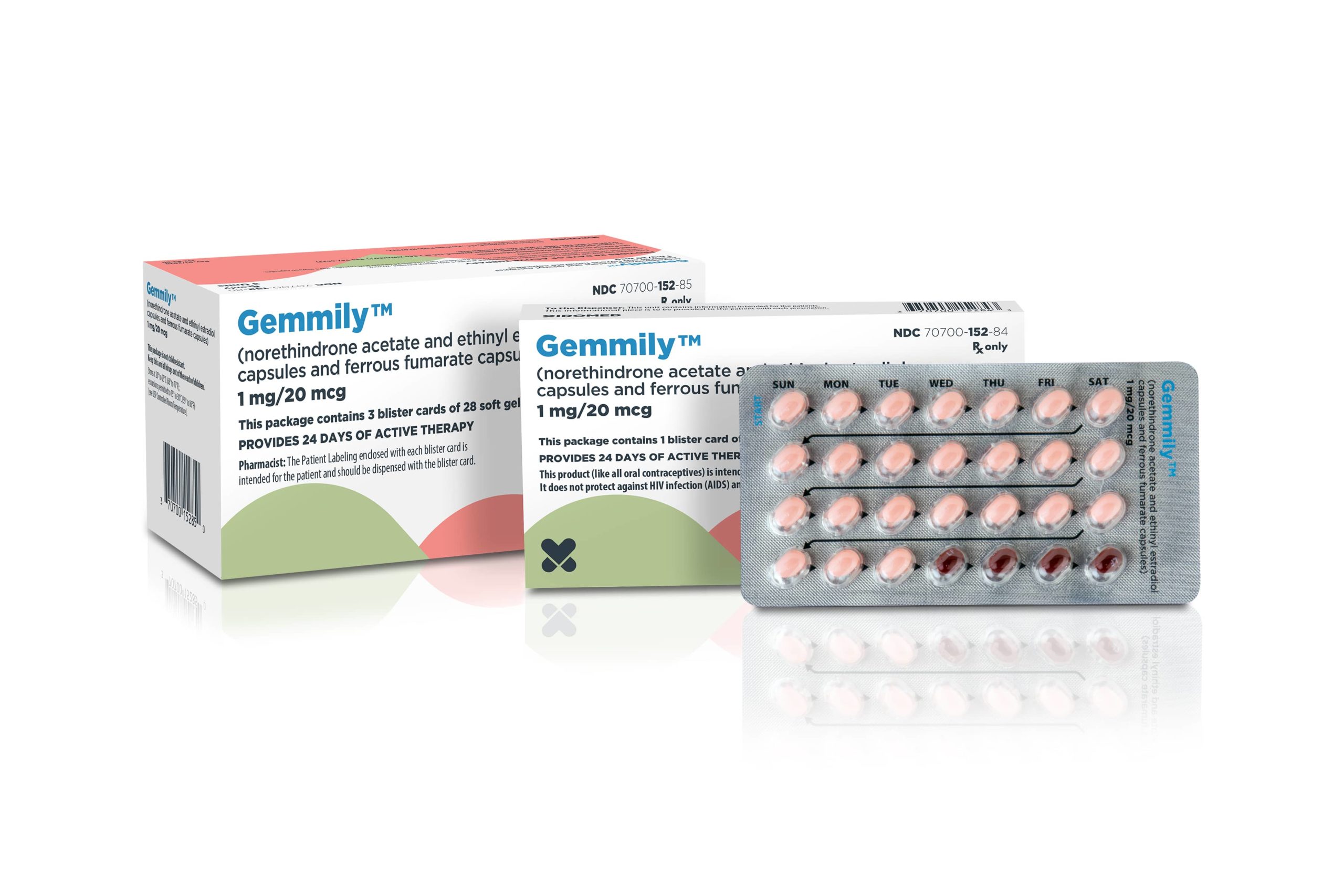
Xiromed LLC, the New Jersey-based generic division of Insud Pharmaceuticals, today announced the FDA approval of Gemmily™ (Norethindrone Acetate and Ethinyl Estradiol Capsules and Ferrous Fumarate Capsules), 1 mg/20 mcg, a therapeutically equivalent generic version of Taytulla®. Xiromed is the first generic to Taytulla® approved in the United States. Commercialization activities for Gemmily™ will begin immediately.
Xiromed CEO, Narasimhan Mani commented, “The launch of this first-to-market generic is an important milestone for Xiromed, demonstrating the strength of our development pipeline, and further establishing our position as a leading generic pharmaceutical company in the women’s health segment. The Xiromed team is excited to offer this first generic product, furthering our commitment to providing high quality generics to patients in the US.”
Taytulla® is an estrogen/progestin combination oral contraceptive indicated for use by women to prevent pregnancy. Annual market sales for Taytulla® for the twelve month period ending August, 2020 were $157 million, according to IQVIA™.
Xiromed LLC, located in Florham Park, NJ, is the US generic division of Insud Pharma, S.L., a global pharmaceutical group headquartered in Madrid, Spain. Xiromed is focused on developing and commercializing high quality generic pharmaceutical products for the US market. In addition to its commercial portfolio of generics available in the US, Xiromed has a robust development portfolio of generic pharmaceutical products in various stages of development, including injectable, inhalation and complex generic products. Learn more at http://www.xiromed.com/usa/



